Why do lemon batteries work?
And chemistry, I guess
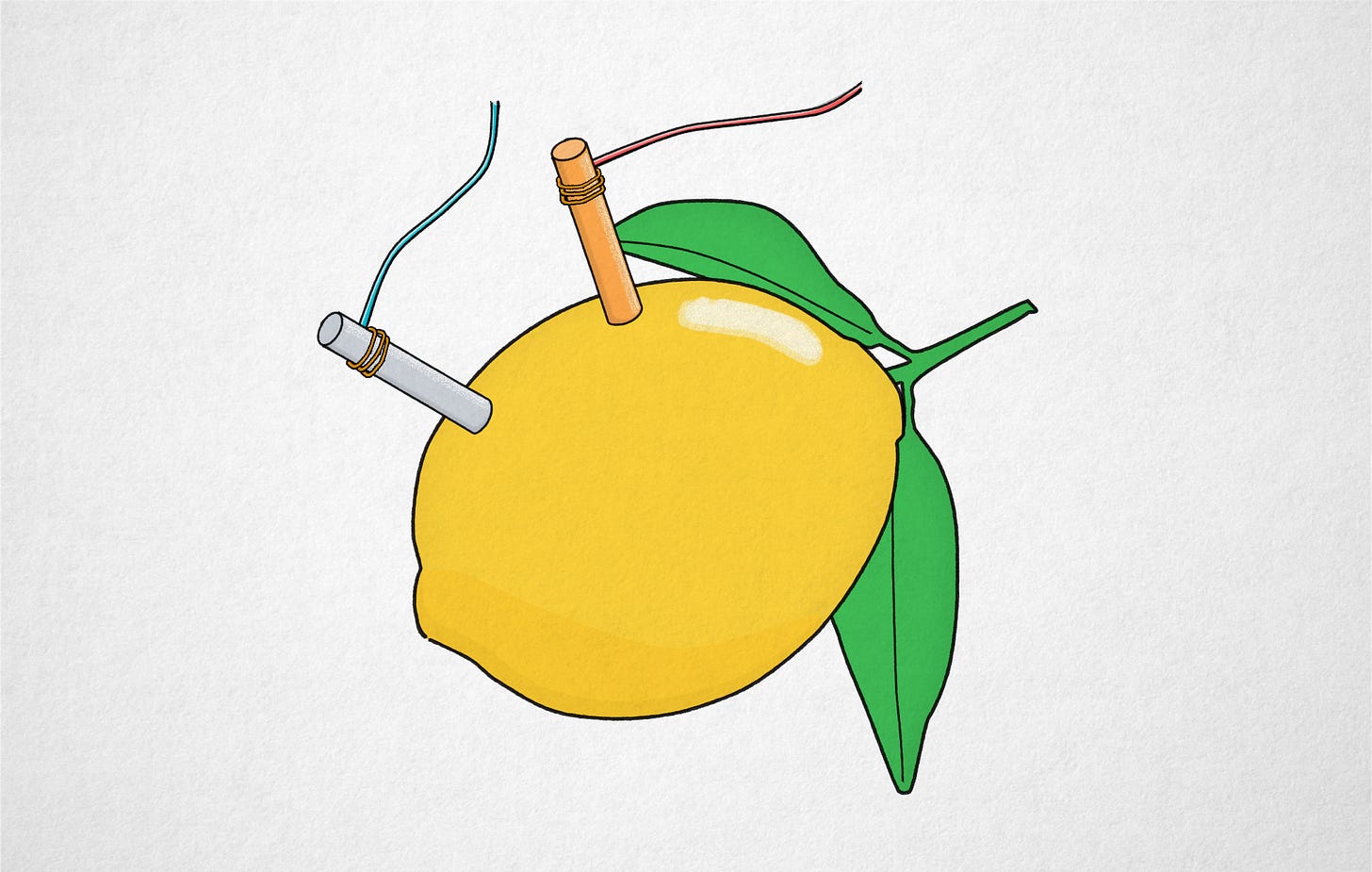
Making a lemon battery is the staple of middle school science activities. The experiment involves jamming two dissimilar metals into a lemon; copper and zinc are the usual picks. If you connect a voltmeter to the electrodes, it should read about 1 V. String two or three lemons in series and you can even light up a small LED.
The chemistry of the lemon cell appears simple, up to a point. Lemon juice is essentially a 5% solution of citric acid in water. Water is a small and strongly polar molecule, and as you probably remember from school, it can cleave the ionic bonds of many substances that were formed by the exchange of electrons and that are held together by the resulting electrostatic field.
This process — known as dissociation — produces electrically-charged halves that lead independent lives in the solution, but revert back to complete molecules once the solvent is removed. In the case of citric acid, we end up with some modest number of positively-charged hydronium cations and negatively-charged citrate anions floating around.
Dude, where’s my hydrogen?
Past this point, introductory texts get hand-wavy. What is explained in school is that many metals react with acids in a redox reaction. Conceptually, the reaction involves hydronium ions snagging electrons from the metal and then turning into hydrogen gas; in parallel, electron-deficient atoms of metal turn into positive ions:
The high-school explanation is that the hydronium ions “want” the electrons more, and when they come across each other in a dark alley, the metal doesn’t put up a fight.
Yet, phrased this way, it’s an electrically-neutral exchange, so it doesn’t quite explain the behavior of a battery, where some electrons are evidently expelled on one terminal and then accepted back on another.
Just as important, the story doesn’t add up for zinc in dilute citric acid: the metal isn’t readily attacked by lemon juice and starts dissolving into Zn2+ cations at an appreciable rate only after an electrical connection between the two terminals is made. Even more confusingly, when the electrodes are connected together, the bulk of the hydrogen starts evolving on the copper electrode, not on the zinc one.
It would appear that in a closed circuit, zinc atoms start spontaneously falling off the electrode, leaving two electrons behind; there are no hydronium ions robbing them at knifepoint. After that, the electrons start moving through a wire toward the copper electrode, where they are handed over to hydronium ions that then morph into hydrogen gas. Why?
The first half of the answer is that is the reduction of the hydronium ion doesn’t happen easily on the surface of zinc. The actual acid-metal reaction is a multi-step process that involves atomic hydrogen diffusing into the metal lattice; zinc is a poor substrate for that. In electrochemistry, we have the concept of overpotential — excess force (i.e., externally-applied voltage) that’s needed to carry out the reaction, compared to what’s suggested by simple thermodynamics. When you measure this force, you find out that the reduction to atomic hydrogen proceeds with less effort on a copper electrode.
It follows that in an open circuit, some zinc atoms may enter the solution and leave electrons behind, but the effect is necessarily self-limiting. This is because the resulting negative charge begins to hinder the process before the voltage rises high enough to allow efficient electron transfer from zinc to H+ ions, which is the only thing that could reduce the accumulated charge.
But what if there is an electrical connection to the copper electrode? Well, electrons can scuttle along the wire with little effort, so the electrostatic field on the Zn side and the Cu side will more or less equalize. And the accumulated negative charge that wasn’t enough to form hydrogen on the zinc side is more than enough to facilitate the reaction on the copper side. Thus, the charge will be dissipated and more Zn2+ ions will form. A sustained current will flow.
The crooked equilibrium
A more fundamental question is why do these reactions happen in the first place. Electrostatic fields are quite powerful; why would an atom ever part with its electrons and then wander off if it’s not being held at gunpoint? Doesn’t that add energy to the system? Why do the properties of zinc and copper differ? How can mirrors be real if our eyes aren't real?!
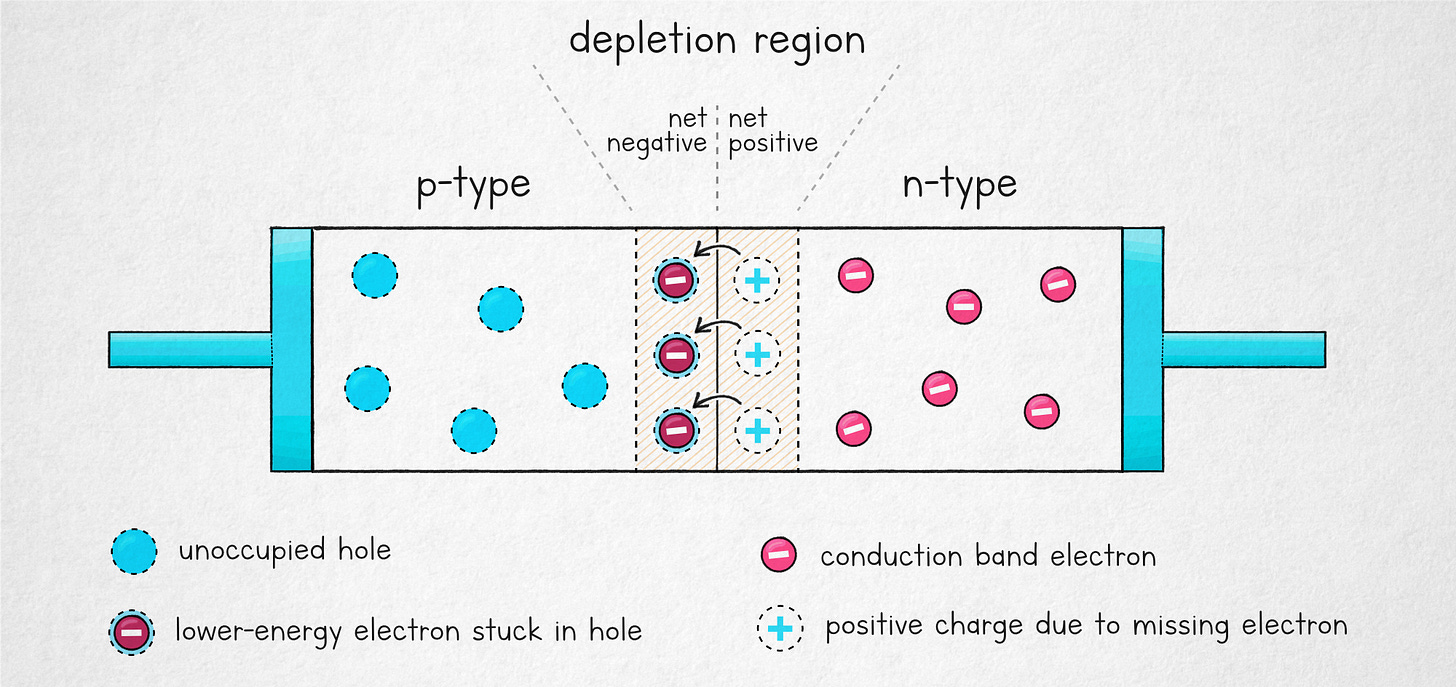
All good questions! If you’re familiar with electronics, a good analogy might be the p-n junction in semiconductors. If you’re not up to speed on the concept, I recommend starting with this article, but in brief: we start by manufacturing an “n-type” material that has some energetic, mobile electrons in a high-energy conduction band. We bring it in contact with a “p-type” material, where electrons occupy a lower-energy valence band, and where there are some available vacancies (holes) at that lower energy level.
Both of the materials are electrically-neutral, but when we bring them together, something interesting happens: a number of the higher-energy electrons from the n-side falls into the lower-energy vacancies on the p-side. This is somewhat akin to a billiard ball falling into a pocket (and staying there). The result is a region with an unbalanced distribution of charges and a built-in electrostatic field:
Similarly to the case of the lemon battery, the effect is self-limiting: eventually, the field grows strong enough that the net inflow of electrons must stop. But the bottom line is that despite the creation of a field, the situation at the junction corresponds to a lower-energy state. If you connect a voltmeter across, it will read zero volts: there are no electrons itching to go back where they used to be.
Semiconductor junctions are not special. Quantum-mechanical properties of molecules and crystal lattices often result in energetically-favorable spots that can be taken up by electrons “belonging” to some other substance. This usually doesn’t result in macroscopic voltages or currents because the process can’t go on for long before the available lower-energy positions are exhausted or the field grows too strong. That said, in the case of batteries, lower-energy products are continuously removed (as gas or ions), and higher-energy reagents are continuously exposed. If there is a conductive pathway to equalize the resulting electric field, the process can go on until the entire electrode or the electrolyte is spent.
In fact, here’s a surprising tidbit: some migration of charges happens spontaneously if you simply bring two dissimilar metals together; no electrolyte is needed at all. The resulting electrostatic field — known as the Volta potential — can be measured with carefully-designed experiments in a vacuum. The importance of this field in guiding electrochemical reactions is apparently not a settled matter in battery science. Some think it’s a key factor; others arrive at similar results from more abstract principles.
👉 For further articles about electronics, click here.
I write well-researched, original articles about geek culture, electronic circuit design, algorithms, and more. If you like the content, please subscribe.

Here are two worthwhile followup articles if you want to get deeper into the weeds:
https://core.ac.uk/download/pdf/215692177.pdf
https://pubs.acs.org/doi/10.1021/acs.jchemed.8b00479
Both are available for free, so no need to work around paywalls.